By Shein-Chung Chow
The U.S. foodstuff and Drug Administration's report back to the country in 2004 and 2005 indicated that one of many most sensible purposes for drug bear in mind used to be that balance info didn't aid current expiration dates. Pharmaceutical businesses behavior balance experiences to symbolize the degradation of drug items and to estimate drug shelf existence. Illustrating how balance stories play a tremendous function in drug defense and caliber insurance, Statistical layout and research of balance experiences offers the foundations and methodologies within the layout and research of balance studies.
After introducing the fundamental thoughts of balance trying out, the booklet makes a speciality of temporary balance stories and experiences numerous tools for estimating drug expiration courting sessions. It then compares a few as a rule hired learn designs and discusses either fastened and random batch statistical analyses. Following a bankruptcy at the statistical equipment for balance research less than a linear combined results version, the publication examines balance analyses with discrete responses, a number of parts, and frozen drug items. furthermore, the writer offers statistical tools for dissolution trying out and explores present matters and up to date advancements in balance studies.
To make sure the defense of shoppers, execs within the box needs to perform balance experiences to figure out the reliability of drug items in the course of their expiration interval. This ebook presents the fabric invaluable that you can practice balance designs and analyses in pharmaceutical learn and improvement
Read or Download Statistical Design and Analysis of Stability Studies PDF
Similar pharmacy books
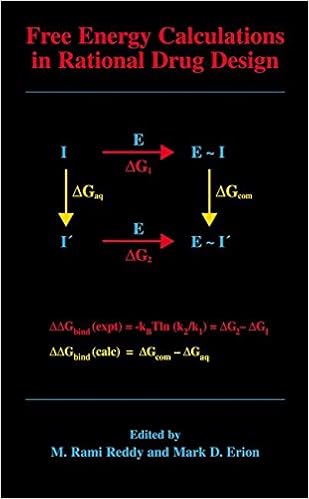
Free Energy Calculations in Rational Drug Design
Unfastened strength calculations signify the main exact computational process to be had for predicting enzyme inhibitor binding affinities. Advances in machine strength within the Nineteen Nineties enabled the sensible program of those calculations in purpose drug layout. This ebook represents the 1st accomplished evaluate of this growing to be sector of study and covers the elemental idea underlying the strategy, a number of cutting-edge options designed to enhance throughput and dozen examples in which loose power calculations have been used to layout and assessment capability drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The college of Pharmacy, college of London: medications, technology and Society, 1842-2012 represents the wealthy historical past of the collage of London tuition of Pharmacy via various colour pictures, very important advances within the pharmacy occupation, cultural milestones, biographies and extra. Written in an enticing and authoritative variety, this e-book depicts the chronological heritage of the college from its institution in 1842 to the current day with a nod towards its aspirations for the long run.
This quantity offers the reader with fresh advances within the fields of molecular toxinology, the biotechnological functions of venom pollutants, and antivenom construction. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the large growth made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous platforms.
Pharmaceutical Process Scale-Up
Concentrating on clinical and useful facets of technique scale-up, this source info the speculation and perform of shifting pharmaceutical approaches from laboratory scale to the pilot plant and creation scale. It covers parenteral and nonparenterel drinks and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress functions, compaction and tableting, and picture coating and regulatory requisites for scale-up and postapproval adjustments.
Extra info for Statistical Design and Analysis of Stability Studies
Example text
4 Stability Analysis with Random Batches To establish an expiration dating period, the 1987 FDA stability guideline requires that at least three batches, and preferably more, be tested in stability analysis to account for batch-to-batch variation so that a single shelf-life is applicable to all future batches manufactured under similar circumstances. , all batches have the same shelf-life), the single shelf-life can be determined based on the ordinary least squares method as the time point at which the 95% confidence bound for the mean degradation curve of the drug characteristics intersects the approved lower specification limit.
34) where tα,J −1 is the αth upper quantile of a central t distribution with J − 1 degrees of freedom. If one is interested in examining whether the reaction rate is different from zero, the following hypotheses should be tested: H0 : βi (h) = 0 vs H0 : βi (h) = 0. 36) where Fα,1,J −1 is the αth upper quantile of a central F distribution with 1 and J − 1 degrees of freedom. We have illustrated the application of a simple linear regression model without intercept through the least-squares method for the estimation of a rate constant for each combination of time points and temperature.
In the next section we describe briefly some deterministic chemical kinetic models. The Arrhenius equation is also described in this section. 2. 3 covers determination of the order of a reaction by selection of adequate models. Also included in this section are issues that often occur in the design of accelerated stability testing. 4. 5. 1 Chemical Kinetic Reaction In this section we describe some functional relationships of chemical kinetic reactions. Unless otherwise stated, all quantities and equations considered in this chapter will be deterministic.