By Dr. med. Dr. phil. Gerhard Nahler (auth.)
The dictionary includes a variety of phrases usually utilized in pharmaceutical drugs. The second variation displays the expanding significance of this technology and the altering regulatory setting particularly on study and improvement of latest remedies in addition to at the behavior of scientific trials, advertising authorisation of latest medicinal items and defense facets together with pharmacovigilance.
The variety of key terms has been significantly enlarged and elevated to over 1,600 phrases; it comprises new medical parts resembling gene remedy and proteomics. moreover, given the significance of the net, the recent variation incorporates a record of most vital websites. just like the first variation, additionally the e-book explains approximately 1,000 abbreviations most typically utilized in pharmaceutical medicine.
This e-book might be a invaluable device for execs within the zone of the pharmaceutical undefined, scientific and pre-clinical study, regulatory affairs, advertising and marketing and advertising authorisation of pharmaceuticals.
Read Online or Download Dictionary of Pharmaceutical Medicine PDF
Best pharmacy books
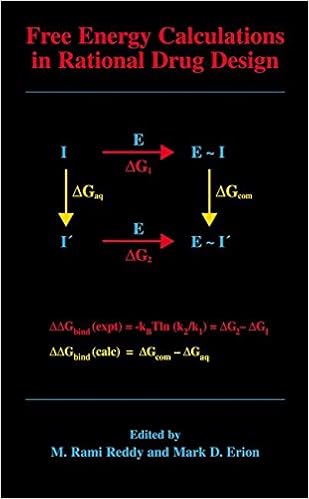
Free Energy Calculations in Rational Drug Design
Unfastened power calculations symbolize the main actual computational approach to be had for predicting enzyme inhibitor binding affinities. Advances in laptop energy within the Nineties enabled the sensible program of those calculations in reason drug layout. This ebook represents the 1st complete evaluate of this growing to be region of study and covers the fundamental thought underlying the strategy, quite a few state-of-the-art innovations designed to enhance throughput and dozen examples in which unfastened power calculations have been used to layout and evaluation power drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The varsity of Pharmacy, collage of London: medicinal drugs, technological know-how and Society, 1842-2012 represents the wealthy historical past of the college of London tuition of Pharmacy via quite a few colour photos, vital advances within the pharmacy occupation, cultural milestones, biographies and extra. Written in an attractive and authoritative kind, this publication depicts the chronological historical past of the varsity from its institution in 1842 to the current day with a nod towards its aspirations for the longer term.
This quantity presents the reader with contemporary advances within the fields of molecular toxinology, the biotechnological functions of venom pollution, and antivenom creation. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the big growth made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous platforms.
Pharmaceutical Process Scale-Up
Targeting medical and sensible points of procedure scale-up, this source info the idea and perform of moving pharmaceutical methods from laboratory scale to the pilot plant and creation scale. It covers parenteral and nonparenterel beverages and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress purposes, compaction and tableting, and picture coating and regulatory necessities for scale-up and postapproval adjustments.
Additional resources for Dictionary of Pharmaceutical Medicine
Sample text
Westport, Conn: Greenwood; 1988:219–223. (ERIC Document) Fuss-Reineck M. Sibling Communication in Star Trek: The Next Generation: Conflicts Between Brothers. Miami, Fla: Annual Meeting of the Speech Communication Association; 1993. ERIC Document Reproduction Service ED364932. (Website) Lynch T. DSN trials and tribble-ations review. Psi Phi: Bradley’s Science Fiction Club Web site. 1996. htm. Accessed October 8, 1997. (Journal Article on the Internet) McCoy LH. Respiratory changes in Vulcans during pon farr.
Concomitant medication Medication taken during treatment with a test drug; → see also drug channelling. condition (EU) In the context of orphan drugs, c. is defined as “any deviation(s) from the normal structure or function of the body, as manifested by a characteristic set of signs and symptoms (typically a recognised distinct disease or a syndrome)”; → see also disease, illness. conditional approval (EU) syn. g. a. may be applied for by the sponsor if: (i) the indications for which the product in question is intended are encountered so rarely that the applicant cannot reasonably be expected to provide comprehensive evidence; (ii) in the present state of scientific knowledge comprehensive information cannot be provided; (iii) it would be contrary to generally accepted principles of medical ethics to collect such information.
G. survival). central ethics committee syn. g. g. c. is sufficient. ); presentation to the european medicines evaluation agency (EMEA) and CPMP resp. is undertaken by the company, the rapporteur member state will be appointed by the CPMP after discussion with the company; the CPMP has 210 days for examination and to reach its opinion which is then send to the Commission, member states and the applicant, including the assessment report, the summary of product characteristics, the labelling and the package insert; the Commission has then 30 days for decision, after which the member states have 28 days for raising questions; then the application goes to the standing committee (with representatives from the member states) and becomes a final Commission’s decision if the majority is in favour; the total time to approval should be max.