By Stanley Nusim
This consultant bargains present and well timed discussions of the method improvement cycle, layout engineering, the approval strategy, qc and coverage, and validation, in addition to plant production actions together with fabrics administration, upkeep, and security.
Read or Download Active Pharmaceutical Ingredients. Development, Manufacturing, and Regulation PDF
Similar pharmacy books
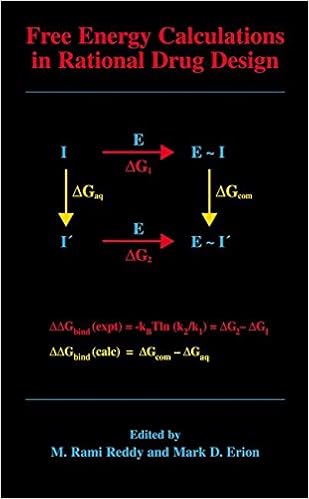
Free Energy Calculations in Rational Drug Design
Loose strength calculations signify the main exact computational procedure on hand for predicting enzyme inhibitor binding affinities. Advances in laptop strength within the Nineteen Nineties enabled the sensible software of those calculations in motive drug layout. This publication represents the 1st finished evaluation of this becoming zone of analysis and covers the fundamental idea underlying the tactic, quite a few cutting-edge thoughts designed to enhance throughput and dozen examples in which unfastened strength calculations have been used to layout and evaluation capability drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The college of Pharmacy, collage of London: medications, technology and Society, 1842-2012 represents the wealthy heritage of the collage of London college of Pharmacy via a variety of colour images, very important advances within the pharmacy occupation, cultural milestones, biographies and extra. Written in an enticing and authoritative kind, this booklet depicts the chronological historical past of the college from its institution in 1842 to the current day with a nod towards its aspirations for the long run.
This quantity presents the reader with contemporary advances within the fields of molecular toxinology, the biotechnological purposes of venom pollutants, and antivenom creation. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the big development made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous structures.
Pharmaceutical Process Scale-Up
Targeting medical and useful points of method scale-up, this source information the speculation and perform of shifting pharmaceutical methods from laboratory scale to the pilot plant and creation scale. It covers parenteral and nonparenterel beverages and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress functions, compaction and tableting, and movie coating and regulatory requisites for scale-up and postapproval adjustments.
Extra info for Active Pharmaceutical Ingredients. Development, Manufacturing, and Regulation
Sample text
There is, of course, no suggestion of the work being completed. Far from it, the consolidation stage is intense in a different way that the development stage was. A great deal of the work ahead is filling blanks (few if the prior work has been done well), refining pilot plant procedures and catching up on the documentation that will support the dossier. Also, the final work on the definition and achievement of the bulk attributes needs to be done to support the final work on the dosage form side and the biobatch and stability studies that will follow.
The Technology Transfer Stage Most of the discussion on the nature and scope of the technology transfer activity is presented in Chapter 3. Nevertheless, the following seems pertinent at this point, as it relates to the technology transfer burden that the bulk process development team carries in addition to its duties on the drug development program: a. A finite effort, even if the midst of a very difficult development stage, must be allocated to look ahead 38 Rosas Figure 13 The consolidation stage in the know-how vs.
12. 3. The Consolidation Stage Although it is not infrequent for a significant bulk process ‘‘loose end’’ to remain tenaciously loose until late in the cycle, by and large the development cycle reaches a stage at which the more difficult development work has been done. To wit: a. The chemical synthesis route is fully defined, albeit sources and specifications of starting materials may still be under negotiation or definition. b. The actual process based on the synthesis route is sufficiently defined and sound pilot plant operating procedures exist or are clearly in the offing.