By Satinder Ahuja, Henrik Rasmussen
Excessive strain, or excessive functionality, liquid chromatography (HPLC) is the tactic of selection for checking purity of recent drug applicants, tracking adjustments in the course of scale up or revision of man-made approaches, comparing new formulations, and working control/assurance of the ultimate drug product. HPLC strategy improvement for prescribed drugs presents an intensive evaluate of contemporary HPLC technique improvement that addresses those distinct matters. features a overview and replace of the present cutting-edge and technological know-how of HPLC, together with idea, modes of HPLC, column chemistry, retention mechanisms, chiral separations, glossy instrumentation (including ultrahigh-pressure systems), and pattern instruction. Emphasis has been put on implementation in a pharmaceutical atmosphere and on delivering a pragmatic perspective.HPLC process improvement for prescription drugs is meant to be quite beneficial for either beginner and skilled HPLC process improvement chemists within the pharmaceutical and for managers who're trying to replace their knowledge.* Covers the necessities for HPLC in a pharmaceutical atmosphere together with thoughts for software program and validation to permit to be used in a regulated laboratory* presents an summary of the pharmaceutical improvement strategy (clinical levels, chemical and pharmaceutical improvement activities)* Discusses how HPLC is utilized in each one part of pharmaceutical improvement and the way tools are constructed to help actions in every one section
Read Online or Download HPLC Method Development for Pharmaceuticals PDF
Similar pharmacy books
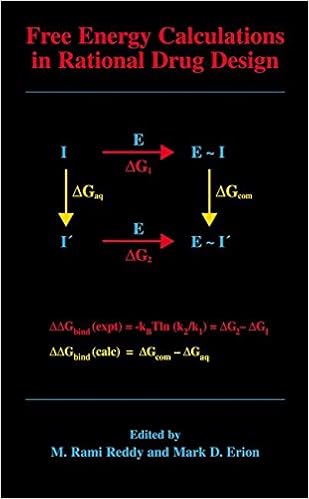
Free Energy Calculations in Rational Drug Design
Unfastened power calculations characterize the main exact computational procedure to be had for predicting enzyme inhibitor binding affinities. Advances in laptop energy within the Nineteen Nineties enabled the sensible program of those calculations in intent drug layout. This booklet represents the 1st complete overview of this becoming region of analysis and covers the elemental concept underlying the tactic, quite a few cutting-edge techniques designed to enhance throughput and dozen examples in which unfastened strength calculations have been used to layout and review strength drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The college of Pharmacy, collage of London: drugs, technology and Society, 1842-2012 represents the wealthy background of the college of London college of Pharmacy via quite a few colour photos, very important advances within the pharmacy career, cultural milestones, biographies and extra. Written in an attractive and authoritative type, this e-book depicts the chronological heritage of the college from its institution in 1842 to the current day with a nod towards its aspirations for the long run.
This quantity presents the reader with fresh advances within the fields of molecular toxinology, the biotechnological functions of venom pollutants, and antivenom creation. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the big growth made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous platforms.
Pharmaceutical Process Scale-Up
Targeting clinical and sensible features of technique scale-up, this source info the speculation and perform of moving pharmaceutical strategies from laboratory scale to the pilot plant and construction scale. It covers parenteral and nonparenterel beverages and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress functions, compaction and tableting, and picture coating and regulatory specifications for scale-up and postapproval adjustments.
Extra resources for HPLC Method Development for Pharmaceuticals
Sample text
XIV. XV. ABSTRACT INTRODUCTION BASIC CHROMATOGRAPHIC DESCRIPTORS EFFICIENCY RESOLUTION MAIN COMPONENTS OFTHE RETENTION MECHANISM GENERAL COLUMN MASS BALANCE PARTITIONING MODEL ADSORPTION MODEL VOID VOLUME CONSIDERATIONS THERMODYNAMIC RELATIONSHIPS SECONDARY EQUILIBRIA SALT EFFECT EFFECT OF DIFFERENT COUNTERANIONS INCLUSION OF SECONDARY EQUILIBRIA IN THE MASS BALANCE CONCLUSIONS REFERENCES ABSTRACT The behavior of the analyte in HPLC columns is discussed from the point of view of analyte distribution between the adsorbent and the bulk liquid phase.
From Figure 2 it is obvious that the smaller the particles the lower the theoretical plate height and the higher the efficiency. The general form of the shown dependence is known as the Van Deemter function (Equation (16)), which has the following mathematical form B H-A+-+Cv (16) v where v is the linear flow velocity, and A, B, and C are constants for given column and mobile phase. 25- 20 15[... o 10 [] o o o I 1 I 2 I 3 I 4 I 5 I 6 Flow velocity F I G U R E 2 The dependence of the theoretical plate height on the flow velocity for columns packed with particles of different diameter.
Trace and Ultratrace Analysis by HPLC, Wiley, New York. 5. Ahuja, S. (2000). Chiral Separations by Chromatography, Oxford, New York. 6. Ahuja, S. (1997). Chiral Separations: Applications and Technology, American Chemical Society, Washington, DC. 7. , and Kirkland, J. (1979). Introduction to Modern Liquid Chromatography, Wiley, New York. 8. , and Kirkland, J. (1988). Practical HPLC Method Development, Wiley, New York. 9. , and Scypinsky, S. (2001). Handbook of Modern pharmaceutical Analysis, Academic, San Diego.