By John A. Bontempo
This up to the moment reference delineates-in a scientific fashion-the applicable, sequential steps for the formula of secure, potent, strong, and marketable liquid parenteral biopharmaceutical products-covering basics and crucial pathways for every section in addition to its goal, functionality, and relation to different phases within the product improvement approach. Written through specialists at the moment eager about state of the art advances within the pharmaceutical drug undefined, improvement of Biopharmaceutical Parenteral Dosage kinds ·details biopharmaceuticals which are approved or present process medical improvement, together with genetically engineered cellphone and engineered vectors within the fermentation technique ·describes purification and characterization strategies for rDNA therapeutics, discussing different types of unit operations for isolation, purification, and characterization ·considers preformulation and formula necessities, reminiscent of physicochemical houses, drug supply, balance stories courses, deactivation/denaturation routes, collection of suitable excipients, and regulatory compliance ·elucidates fundamentals of analytical strategies, tools improvement, separation equipment utilizing chromatographic and electrophoretic thoughts, and bioactivity tools protecting bioassays and immunoassays for quantifying the steadiness of organic job ·shows the right way to decide on definitely the right filter out for maximizing compatibility and minimizing adsorption and inactivation, analyzing subject matters from easy filtration theories to destiny traits ·reviews the choice procedure for suitable elastomeric closures, interpreting actual, chemical, toxicological houses, protein adsorption on elastomeric surfaces, thoughts to reduce/eliminate adsorption, and really good boxes for biotechnological purposes ·and extra! supplied with priceless references, tables, and drawings, this sensible advisor is imperative for pharmaceutical, medicinal, and protein chemists; molecular biologists; procedure engineers; purification scientists; biopharmaceutical and pharmaceutical formulators and product builders; qc, caliber insurance, and regulatory compliance group of workers; and upper-level undergraduate and graduate scholars in those disciplines.
Read Online or Download Development of biopharmaceutical parenteral dosage forms PDF
Similar pharmacy books
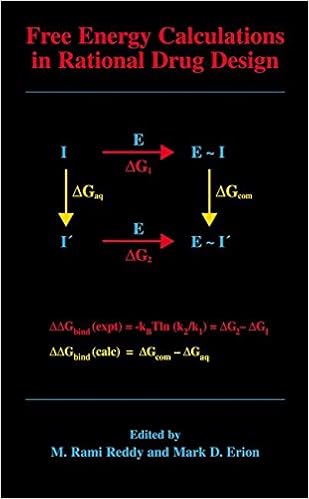
Free Energy Calculations in Rational Drug Design
Unfastened strength calculations characterize the main actual computational approach on hand for predicting enzyme inhibitor binding affinities. Advances in desktop strength within the Nineteen Nineties enabled the sensible software of those calculations in intent drug layout. This e-book represents the 1st entire evaluate of this growing to be zone of analysis and covers the fundamental idea underlying the strategy, quite a few cutting-edge ideas designed to enhance throughput and dozen examples in which loose power calculations have been used to layout and overview strength drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The varsity of Pharmacy, college of London: medications, technology and Society, 1842-2012 represents the wealthy historical past of the college of London university of Pharmacy via quite a few colour pictures, very important advances within the pharmacy career, cultural milestones, biographies and extra. Written in an interesting and authoritative sort, this publication depicts the chronological heritage of the varsity from its institution in 1842 to the current day with a nod towards its aspirations for the long run.
This quantity offers the reader with contemporary advances within the fields of molecular toxinology, the biotechnological purposes of venom pollutants, and antivenom creation. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the large development made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous structures.
Pharmaceutical Process Scale-Up
Targeting clinical and sensible points of method scale-up, this source info the speculation and perform of moving pharmaceutical methods from laboratory scale to the pilot plant and creation scale. It covers parenteral and nonparenterel beverages and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress functions, compaction and tableting, and picture coating and regulatory standards for scale-up and postapproval alterations.
Extra info for Development of biopharmaceutical parenteral dosage forms
Sample text
Bontempo 91 Page viii 5. Formulation Development John A. Bontempo 109 6. The Analytical Techniques Basant G. Sharma 143 7. Membrane Filtration Technology Forrest Badmington 171 8. Considerations for Elastomeric Closures for Parenteral Biopharmaceutical Drugs John A. Bontempo 223 Index 241 Page ix Contributors Forrest Badmington Senior Consulting Scientist, Millipore Corporation, Bedford, Masssachusetts John A. D. Consultant, Biopharmaceutical Product Development, East Brunswick, New Jersey Anthony S.
Aminoglycoside antibiotic resistance genes are commonly used for selecting transformed host cells but different antibiotics are employed depending upon the host chosen (typically, kanamycin or neomycin for E. coli and G418 for animal cells). Occasionally, quality control of the purified biopharmaceutical is enhanced by careful choices of vector components. In several cases, co-purification of other proteins encoded by the expression vectors could be detected in the purified biopharmaceutical and elimination of the contami- Page 18 nant from the product depended upon reengineering the vector to remove the offending gene.
R. Nixon Drug Metabolism: Chemical and Biochemical Aspects, Bernard Testa and Peter Jenner New Drugs: Discovery and Development, edited by Alan A. Rubin Sustained and Controlled Release Drug Delivery Systems, edited by Joseph R. Robinson Modern Pharmaceutics, edited by Gilbert S. Banker and Christopher T. Rhodes Prescription Drugs in Short Supply: Case Histories, Michael A. Schwartz Activated Charcoal: Antidotal and Other Medical Uses, David O. Cooney Concepts in Drug Metabolism (in two parts), edited by Peter Jenner and Bernard Testa Pharmaceutical Analysis: Modern Methods (in two parts), edited by James W.