By Deborah Rosenbaum
This revised variation of a bestseller offers a logical, step by step advisor to checking out new medicines and remedy modalities in compliance with the most recent FDA rules. With present varieties, ICH GCP info, FDA rules, and different references, it exhibits readers find out how to deal with a scientific study research successfully and successfully.
Read or Download Clinical research coordinator handbook: GCP tools and techniques PDF
Best pharmacy books
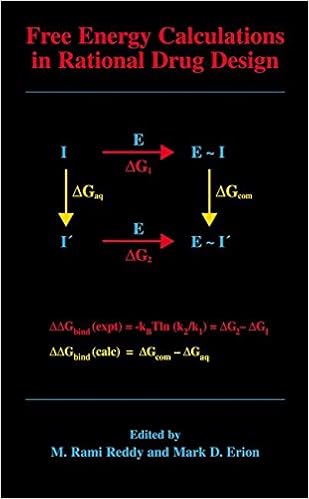
Free Energy Calculations in Rational Drug Design
Loose power calculations characterize the main exact computational process to be had for predicting enzyme inhibitor binding affinities. Advances in desktop energy within the Nineties enabled the sensible program of those calculations in purpose drug layout. This ebook represents the 1st finished assessment of this growing to be quarter of analysis and covers the elemental idea underlying the tactic, various cutting-edge techniques designed to enhance throughput and dozen examples in which unfastened power calculations have been used to layout and review power drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The college of Pharmacy, college of London: medications, technological know-how and Society, 1842-2012 represents the wealthy historical past of the college of London university of Pharmacy via a variety of colour pictures, very important advances within the pharmacy career, cultural milestones, biographies and extra. Written in a fascinating and authoritative type, this publication depicts the chronological historical past of the college from its institution in 1842 to the current day with a nod towards its aspirations for the longer term.
This quantity presents the reader with fresh advances within the fields of molecular toxinology, the biotechnological functions of venom pollutants, and antivenom construction. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the large development made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous platforms.
Pharmaceutical Process Scale-Up
Targeting medical and useful facets of approach scale-up, this source information the idea and perform of shifting pharmaceutical approaches from laboratory scale to the pilot plant and construction scale. It covers parenteral and nonparenterel drinks and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress purposes, compaction and tableting, and picture coating and regulatory standards for scale-up and postapproval alterations.
Additional info for Clinical research coordinator handbook: GCP tools and techniques
Example text
The regulations for informed consent (21 CFR 50 and 45 CFR 46) and IRBs (21 CFR 56) are aimed at protecting the subjects in clinical trials. Part 312 (IND regulations) enforces this policy in the context of a sponsor's submission of an application to the FDA to begin human trials with an Investigational New Drug (IND). Clinical trial data are collected in support of a New Drug Application (NDA), a formal request to the FDA to approve the investigational new drug for marketing for a specific indication based on data collected from clinical trials.
8. 9 Food and Drug Administration Federal Register, 1127191, Vol. S. IND) NIA Federal Register, Vol. 56 (93) (1991 ) Acceptance of Foreign Clinical Studies (9198) Gender NIA Evaluation of Gender Differences (9198) all Federal Register,Vol. 52 (53), (3119187) Preamble and original Rewrite of regulations, Part VII, p. 2 continued from previous page 21 CFR ICH (E6) Information SheetsiReferences* Payment for lnvestigational Products (9198) Payment to Research Subjects (9198) Recruiting Study Subjects (9198) Significant Differences in HHS and FDA Regulations for lRBs and Informed Consent (9198) Waiver of IRB Requirements (9198) Cooperative Research (9198) Nonlocal IRB Review (9198) Expedited Review, Federal Register, Vol.
Lab evaluations, X-rays, office visits) State the route of administration of the experimental agent. 2. Define RISKS attributable to the experimental agent and/or procedures. 3. Discuss any expected BENEFITS from participation in the trial. 4. Discuss ALTERNATIVE TREATMENTS. 5. State the policy for protection of CONFIDENTIALITY of records, noting that a qualified representative of the sponsor and the FDA may inspect subject study records. 6. Discuss whether COMPENSATION for study-related injuries is provided and if EMERGENCY TREATMENT will or will not be provided by the institution.