By Michael R. Bleavins, Claudio Carini, Mallé Jurima-Romet, Ramin Rahbari
Realize how biomarkers can strengthen the luck fee of drug improvement effortsAs pharmaceutical businesses fight to enhance the good fortune expense and cost-effectiveness of the drug improvement approach, biomarkers have emerged as a precious device. This e-book synthesizes and reports the most recent efforts to spot, advance, and combine biomarkers as a key process in translational drugs and the drug improvement method. jam-packed with case reports, the publication demonstrates how biomarkers can increase drug improvement timelines, decrease expenditures, facilitate higher compound choice, lessen late-stage attrition, and open the door to customized medicine.Biomarkers in Drug improvement is split into 8 parts:Part One bargains an summary of biomarkers and their function in drug development.Part highlights very important applied sciences to aid researchers determine new biomarkers.Part 3 examines the characterization and validation method for either medications and diagnostics, and offers useful suggestion on acceptable statistical ways to make sure that biomarkers satisfy their meant purpose.Parts 4 via Six research the appliance of biomarkers in discovery, preclinical security evaluate, scientific trials, and translational medicine.Part Seven specializes in classes realized and the sensible elements of imposing biomarkers in drug improvement programs.Part 8 explores destiny traits and concerns, together with info integration, customized medication, and moral concerns.Each of the thirty-eight chapters was once contributed by means of a number of top specialists, together with scientists from biotechnology and pharmaceutical businesses, academia, and the U.S. nutrition and Drug management. Their contributions supply pharmaceutical and scientific researchers the main up to date figuring out of the techniques used for and purposes of biomarkers in drug improvement.
Read Online or Download Biomarkers in Drug Development: A Handbook of Practice, Application, and Strategy PDF
Similar pharmacy books
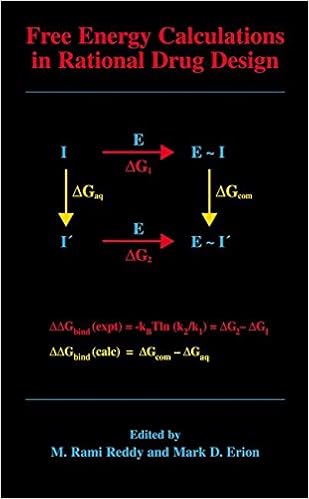
Free Energy Calculations in Rational Drug Design
Unfastened power calculations characterize the main actual computational approach to be had for predicting enzyme inhibitor binding affinities. Advances in laptop strength within the Nineteen Nineties enabled the sensible program of those calculations in cause drug layout. This publication represents the 1st accomplished overview of this becoming region of study and covers the fundamental concept underlying the tactic, a variety of cutting-edge ideas designed to enhance throughput and dozen examples in which loose strength calculations have been used to layout and assessment power drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The college of Pharmacy, college of London: medicinal drugs, technological know-how and Society, 1842-2012 represents the wealthy background of the college of London institution of Pharmacy via various colour images, very important advances within the pharmacy career, cultural milestones, biographies and extra. Written in an interesting and authoritative sort, this booklet depicts the chronological background of the college from its institution in 1842 to the current day with a nod towards its aspirations for the longer term.
This quantity presents the reader with fresh advances within the fields of molecular toxinology, the biotechnological purposes of venom pollution, and antivenom creation. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the big development made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous platforms.
Pharmaceutical Process Scale-Up
Concentrating on clinical and useful features of strategy scale-up, this source information the idea and perform of shifting pharmaceutical approaches from laboratory scale to the pilot plant and creation scale. It covers parenteral and nonparenterel drinks and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress functions, compaction and tableting, and picture coating and regulatory requisites for scale-up and postapproval alterations.
Additional resources for Biomarkers in Drug Development: A Handbook of Practice, Application, and Strategy
Example text
The FDA Critical Path Initiative was developed in 2006, with many opportunities described for advancing biomarkers and surrogate endpoints in a broad range of areas for therapeutic development [19,20]. Progress in these areas have augmented industry knowledge of application of biomarkers in clinical development programs and fostered harmony with international regulatory organizations in the ever-expanding global research environment. This program has been making progress on expanding the toolbox for clinical development—many of the components foster development and application of biomarkers.
FDA (2004). Challenge and opportunity on the critical path to new medical products. html (accessed Aug. 23, 2008). 21. FDA (2008). European Medicines Agency to consider additional test results when assessing new drug safety. gov/bbs/topics/NEWS/2008/NEW01850. html. 22. Woolsey RL, Cossman J (2007). Drug development and the FDA’s Critical Path Initiative. Clin Pharmacol Ther, 81:129–133. REFERENCES 29 23. The Biomarkers Consortium (2008). On the critical path of drug discovery. Clin Pharmacol Ther, 83:361–364.
In 2007, Congress authorized the establishment of a private–public resource to support collaborative research with the FDA. One of the major obstacles to clinical genomic research expressed over the years has been a concern that research participants may be discriminated against in employment and provision of health insurance benefits as a result of the association of genetic disease markers. S. Congress passed legislation known as the Genetic Information Non-discrimination Act of 2008, preventing the use of genetic information to deny employment and health insurance.