By Mark Chasin
Experiences the homes, synthesis, and formulations of a few good studied polymers more and more getting used in site-specific or systematic management of pharmaceutical brokers. for every of the polymers, discusses the historical past; chemistry and synthesis; the formula of microcapsules, solv
Read Online or Download Biodegradable Polymers as Drug Delivery Systems (Drugs and the Pharmaceutical Sciences) PDF
Best pharmacy books
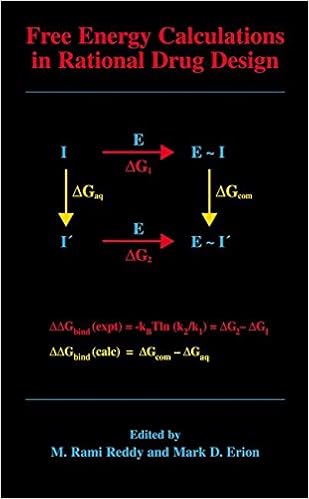
Free Energy Calculations in Rational Drug Design
Loose strength calculations characterize the main exact computational approach to be had for predicting enzyme inhibitor binding affinities. Advances in machine strength within the Nineteen Nineties enabled the sensible program of those calculations in cause drug layout. This ebook represents the 1st entire assessment of this becoming zone of analysis and covers the fundamental idea underlying the strategy, quite a few cutting-edge thoughts designed to enhance throughput and dozen examples in which unfastened power calculations have been used to layout and evaluation power drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The college of Pharmacy, college of London: medications, technological know-how and Society, 1842-2012 represents the wealthy historical past of the college of London institution of Pharmacy via a variety of colour photos, vital advances within the pharmacy occupation, cultural milestones, biographies and extra. Written in an interesting and authoritative variety, this publication depicts the chronological heritage of the varsity from its institution in 1842 to the current day with a nod towards its aspirations for the long run.
This quantity presents the reader with contemporary advances within the fields of molecular toxinology, the biotechnological purposes of venom pollutants, and antivenom creation. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the large growth made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous platforms.
Pharmaceutical Process Scale-Up
Targeting medical and functional elements of approach scale-up, this source information the idea and perform of shifting pharmaceutical methods from laboratory scale to the pilot plant and creation scale. It covers parenteral and nonparenterel drinks and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress purposes, compaction and tableting, and movie coating and regulatory standards for scale-up and postapproval adjustments.
Extra info for Biodegradable Polymers as Drug Delivery Systems (Drugs and the Pharmaceutical Sciences)
Sample text
Significantly higher molecular weights were achieved using shorter reaction times by utilizing cadmium acetate, earth metal oxides, and ZnEt2-H20. The molecular weights ranged from 140,935 to 245,010 with catalysts, in comparison Polyanhydrides as Drug Delivery Systems 45 to the material described above, which had a molecular weight of 116,800 when produced without- catalysts. -toluenesulfonic acid (p-TSA) or basic 4-dimethylaminopyridine (4-DMAP) catalysts were tested, the acid catalyst p-TSA did not show any effect while the basic catalyst (4-DMAP) caused a decrease in molecular weight.
More than 16 days protection was afforded versus on ly 3 days for controls (137). A unique method of formulating delivery systems based on starchl PLA systems was studied (138). In that approach, the goal was to provide a better matrix for delivery of high molecular weight hydrophilic molecules. A hydrophilic material, starch,was combined through graft polymerization to PLA. The carbolactic polymers were then used to entrap bovine serum albumin in microspheres. Considerable effort has been devoted to the development of controlled release formulations for growth hormone, especially porcine and bovine growth hormone.
Tice, T. , Meyers, W. , Schalley, A. , and Redding, T. , Inhibition of rat prostate tumors by controlled release of [D-T rp6] - LHRH from injectable microcapsules, Proc. Int. Symp. Control. ReI. Bioact. , 88, 1984. , Controlled-release of leuprolide acetate from Laet i de / Glycol ide Pol ymers 128. 129. 130. 131. 132. 133. 134. 135. 136. 137. 138. 139. 41 polylactic acid or copoly(lacticl glycolic) acid microcapsules: Influence of molecular weight and copolymer ratio of polymer, Chern. Pharm. , 36, 1502, 1988.