By Edith Mathiowitz, Donald E. Chickering III, Claus-Michael Lehr
This useful reference offers a accomplished overview of the elemental tools for characterizing bioadhesive fabrics and enhancing automobile concentrating on and uptake-offering chances for reformulating present compounds to create new prescription drugs at decrease improvement charges. Evaluates the original service features of bioadhesive polymers and their strength to augment localization of added brokers, neighborhood bioavailability, and drug absorption and delivery! Written by means of over 50 overseas specialists and reflecting huge wisdom of either conventional bioadhesive recommendations and novel medical functions, Bioadhesive Drug supply platforms ·discusses mechanical and chemical bonding, polymer-mucus interactions, the impression of floor strength in bioadhesion, polymer hydration, and mucus rheology ·analyzes biochemical houses of mucus and glycoproteins, cellphone adhesion molecules, and mobile interplay with - and 3-dimensional surfaces ·covers microbalances and magnetic strength transducers, atomic strength microscopy, direct measurements of molecular point adhesions, and techniques to degree cell-cell interactions ·examines bioadhesive companies, diffusion or penetration enhancers, and lectin-targeted autos ·describes vaginal, nasal, buccal, ocular, and transdermal drug supply ·reviews bioadhesive interactions with the mucosal tissues of the attention and mouth, and people within the breathing, urinary, and gastrointestinal tracts ·explores problems with product improvement, medical checking out, and construction ·and extra! Amply referenced with over 1400 bibliographic citations, and illustrated with greater than three hundred drawings, photos, tables, and show equations, Bioadhesive Drug supply structures serves as a valid foundation for innovation in bioadhesive structures and a very good advent to the topic. This particular reference is perfect for pharmaceutical scientists and technologists; chemical, polymer, and plastics engineers; biochemists; actual, floor, and colloid chemists; biologists; and upper-level undergraduate and graduate scholars in those disciplines.
Read or Download Bioadhesive drug delivery systems: fundamentals, novel approaches, and development PDF
Similar pharmacy books
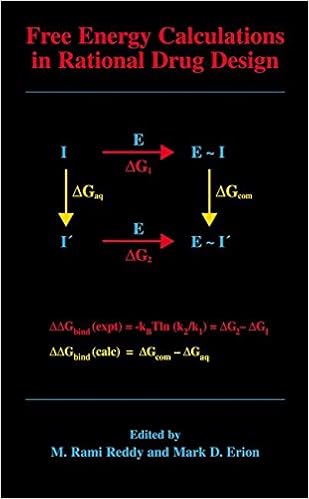
Free Energy Calculations in Rational Drug Design
Unfastened power calculations signify the main exact computational technique on hand for predicting enzyme inhibitor binding affinities. Advances in laptop strength within the Nineties enabled the sensible program of those calculations in reason drug layout. This ebook represents the 1st complete evaluation of this becoming quarter of analysis and covers the fundamental thought underlying the tactic, quite a few state-of-the-art thoughts designed to enhance throughput and dozen examples in which loose power calculations have been used to layout and assessment power drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The varsity of Pharmacy, collage of London: medications, technology and Society, 1842-2012 represents the wealthy historical past of the college of London institution of Pharmacy via a number of colour photos, very important advances within the pharmacy career, cultural milestones, biographies and extra. Written in an enticing and authoritative variety, this ebook depicts the chronological heritage of the college from its institution in 1842 to the current day with a nod towards its aspirations for the long run.
This quantity presents the reader with contemporary advances within the fields of molecular toxinology, the biotechnological purposes of venom pollution, and antivenom creation. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the large growth made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous platforms.
Pharmaceutical Process Scale-Up
Targeting clinical and sensible facets of procedure scale-up, this source info the idea and perform of moving pharmaceutical tactics from laboratory scale to the pilot plant and creation scale. It covers parenteral and nonparenterel drinks and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress purposes, compaction and tableting, and movie coating and regulatory necessities for scale-up and postapproval alterations.
Additional resources for Bioadhesive drug delivery systems: fundamentals, novel approaches, and development
Sample text
4. Optimize. Continuously improve process understanding by monitoring process performance over time and comparing it with design objectives; utilize improved understanding to optimize process operation to ensure consistent attainment of approval specifications for the CQAs. Before discussing how these tasks should be accomplished in practice, it is important to place all four of them within the context of the spirit of and organizing principles behind PAT. For the products of any manufacturing process to meet predefined quality specifications consistently, there must be a comprehensive measurement system in place for acquiring appropriate measurements of key process variables and of raw materials; there must also be a reliable system determining critical quality and performance attributes of end products sufficiently frequently.
The potential benefits are great, for example, real-time release testing leading to a reduction in end-product release testing. Real-time release testing refers to the ability to evaluate and ensure the acceptable quality of in-process and/or final product based on process data. PAT could also result in elimination of superfluous monitoring and control at the manufacturing scale (Dean and Bruttin 2000). 4 conclusIons The complicated nature of biotech products makes understanding and controlling their production process not just necessary but often times difficult.
A post-production, off-line quality control strategy makes biopharmaceutical manufacturing processes extremely inefficient because the only reasonable course of action when a CQA is found to be out of range is to discard the entire product batch (due to regulatory concerns). Thus, even when the design space has been properly established, meeting CQA specifications consistently (and efficiently) with current industrial quality control procedures remains a major challenge. On-line product quality control, which has been a standard in the petroleum and chemical process industries for decades (Schuyten 1967; Ogunnaike 1994), significantly improves process efficiency because it makes “in-production” corrections possible, thereby preventing out-of-specification conditions from persisting during production.