By Harry G. Brittain
Volumes during this greatly respected sequence current accomplished studies of drug ingredients and extra fabrics, with severe evaluation chapters that summarize details regarding the characterization of drug ingredients and excipients. This organizational constitution meets the desires of the pharmaceutical neighborhood and allows the advance of a well timed car for publishing assessment fabrics in this subject. The scope of the Profiles sequence encompasses evaluation articles and database compilations that fall inside one of many following six huge different types: actual profiles of drug components and excipients; Analytical profiles of drug components and excipients; Drug metabolism and pharmacokinetic profiles of drug ingredients and excipients; technique relating to the characterization of drug ingredients and excipients; tools of chemical synthesis; and studies of the makes use of and functions for person drug elements, periods of drug components, or excipients. * offers finished stories overlaying all facets of drug improvement and formula of substances * Profiles creatine monohydrate and fexofenadine hydrochloride, in addition to 5 others * Meets the knowledge wishes of the drug improvement group. learn more... summary: Volumes during this commonly respected sequence current complete reports of drug components and extra fabrics, with severe evaluate chapters that summarize info relating to the characterization of drug components and excipients. This organizational constitution meets the desires of the pharmaceutical group and makes it possible for the advance of a well timed car for publishing assessment fabrics in this subject. The scope of the Profiles sequence encompasses overview articles and database compilations that fall inside one of many following six vast different types: actual profiles of drug elements and excipients; Analytical profiles of drug ingredients and excipients; Drug metabolism and pharmacokinetic profiles of drug elements and excipients; method on the topic of the characterization of drug components and excipients; tools of chemical synthesis; and studies of the makes use of and purposes for person drug components, periods of drug components, or excipients. * offers complete studies overlaying all points of drug improvement and formula of gear * Profiles creatine monohydrate and fexofenadine hydrochloride, in addition to 5 others * Meets the data wishes of the drug improvement group
Read Online or Download Analytical Profiles of Drug Substances, Excipients, and Related Methodology PDF
Best pharmacy books
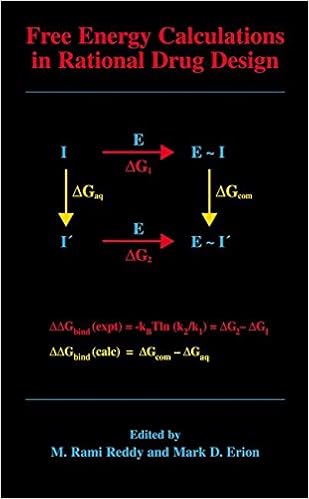
Free Energy Calculations in Rational Drug Design
Loose strength calculations signify the main exact computational technique to be had for predicting enzyme inhibitor binding affinities. Advances in laptop strength within the Nineteen Nineties enabled the sensible program of those calculations in purpose drug layout. This booklet represents the 1st complete evaluation of this turning out to be sector of analysis and covers the elemental conception underlying the strategy, a variety of state-of-the-art suggestions designed to enhance throughput and dozen examples in which unfastened strength calculations have been used to layout and overview power drug applicants.
The School of Pharmacy, University of London. Medicines, Science and Society, 1842–2012
The college of Pharmacy, collage of London: medications, technological know-how and Society, 1842-2012 represents the wealthy background of the college of London university of Pharmacy via a variety of colour images, vital advances within the pharmacy occupation, cultural milestones, biographies and extra. Written in an enticing and authoritative sort, this ebook depicts the chronological background of the college from its institution in 1842 to the current day with a nod towards its aspirations for the long run.
This quantity presents the reader with fresh advances within the fields of molecular toxinology, the biotechnological functions of venom pollution, and antivenom creation. The content material of the twenty chapters of Venom Genomics and Proteomics illustrates not just the large development made because the implementation of omics applied sciences within the box of toxinology, yet one additionally realizes the line nonetheless forward to arrive a holistic view of venomous platforms.
Pharmaceutical Process Scale-Up
Targeting clinical and functional features of strategy scale-up, this source info the speculation and perform of shifting pharmaceutical tactics from laboratory scale to the pilot plant and creation scale. It covers parenteral and nonparenterel drinks and semi-solids, items derived from biotechnology, dry mixing and powder dealing with, granulation and drying, fluid mattress purposes, compaction and tableting, and movie coating and regulatory requisites for scale-up and postapproval alterations.
Additional info for Analytical Profiles of Drug Substances, Excipients, and Related Methodology
Example text
450. [32] H. H. , Inc, Brooklyn, New York, 1943, p. 49. [33] M. Wyss and R. Kaddurah-Daouk, Physiol. , 2000, 80, 1107–1213. , ‘‘Evaluated Infrared Reference Spectra’’ in NIST Chemistry WebBook, NIST Standard Reference Database Number 69, Eds. P. J. Linstrom and W. G. Mallard, June 2005, National Institute of Standards and Technology, Gaithersburg MD, p. gov). [35] A. L. Smith, The coblentz society desk book of infrared spectra, in The Coblentz Society Desk Book of Infrared Spectra (ed. C. D. , The Coblentz Society, Kirkwood, MO, 1982, pp.
Jenkins, K. L. Ferrante, M. Thomas, A. Friedliche, S. E. Brownee, G. Schillingg, D. R. Borcheltg, S. M. Herschh, C. A. Rossi, et al. Neurobiol. , 2001, 8, 479–491. [14] A. Bender, D. P. Auer, T. Merl, R. Reilmann, P. Saemann, A. Yassouridis, J. Bender, A. Weindl, M. Dose, T. Gasser and T. Klopstock, J. , 2005, 252, 36–41. [15] A. C. Ellis and J. Rosenfeld, CNS Drugs, 2004, 18, 967–980. [16] L. Mazzini, C. Balzarini, R. Colombo, G. Mora, I. Pastore, R. De Ambrogio and M. Caligari, J. Neurol. , 2001, 191, 139–144.
1985, 23, 371–376. [56] Y. Yokoyama, S. Tsuji and H. Sato, J. Chromatogr. A, 2005, 1085, 110–116. [57] P. Kennedy, Biol. Advertising Suppl. The Application Note book, Leco Corporation, 2006, 24. [58] P. Fossati, L. Prenclpe and G. Bert, Clin. , 1963, 29, 1494–1496. [59] J. A. Weber and A. P. van Zanten, Clin. , 1991, 37, 695–700. [60] A. K. Dash, D. W. Miller, H. Y. Han, J. Carnazzo and J. R. Stout, J. Pharm. , 2001, 90, 1593–1598. [61] S. Ganguly, S. Jayappa and A. K. Dash, AAPS Pharm. Sci. , 4, Article 26.